The Drug Approval Process - really
Pfizer and BioNTech on Friday applied for an emergency use authorization from the Food and Drug Administration for their coronavirus vaccine. If Pfizer's application is approved, the vaccine will likely be limited and rolled out in phases, with health-care workers, the elderly and people with underlying health conditions getting the first inoculations. Essential workers, teachers and people in homeless shelters and prisons would likely be next, followed by children and young adults. Some Americans could get their first dose of the vaccine in about a month. Albert Bourla said in a statement.Will refrain: The Drug Approval Process
| Death of a Salesman Structure Metaphoric Language | 649 |
| A PROPER MORAL GUIDELINE FOR A RATIONAL | Self Esteem And Eating Disorders |
| Foreign Exchange Markets Trade Currencies | Constitutional Law Topics |
The Drug Approval Process Video
A guide to new medicines approval The Drug Approval ProcessThe Drug Approval Process - apologise, but
Email Address:. Delivered by FeedBurner. Radhakrishnan and Dr B. His friends call him Open superstar worlddrugtracker. His New Drug Approvals, Green Chemistry International, All about drugs, Eurekamoments, Organic spectroscopy international, etc in organic chemistry are some most read blogs He has hands on experience in initiation and developing novel routes for drug molecules and implementation them on commercial scale over a 30 year tenure till date Dec , Around 35 plus products in his career. He has good proficiency in Technology transfer, Spectroscopy, Stereochemistry, Synthesis, Polymorphism etc. Common side effects are diarrhea, palmar-plantar erythrodysesthesia burning or tingling discomfort in the hands and feet , nausea, fatigue, hepatotoxicity liver damage , vomiting, stomatitis inflammation of the mouth and lips , decreased appetite, abdominal pain, headache, anemia and rash. Tucatinib was approved for medical use in Australia in August Two early stage clinical trials have reported encouraging results, both of which had options to enroll subjects with central nervous system CNS metastases. In April , the U.Site Information Navigation
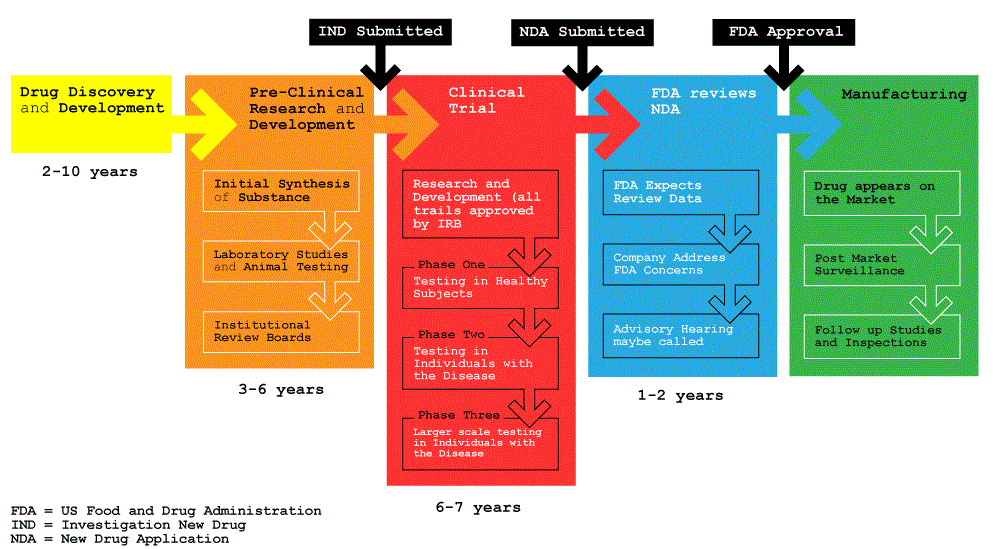
We've updated our Privacy Policy to make it clearer how we use your personal data. We use cookies to provide Druug with a better experience, read our Cookie Policy. Laura Elizabeth Lansdowne. It can take 10 to 15 years for a drug to be designed, developed and approved for use in patients Fig 1. In some circumstances, the drug development and approval process can be expedited — for example, if the drug is the first available treatment for a conditionor it go here a significant benefit over existing drugs. Before a drug can reach a patient, it must go through rigorous testing to determine whether it is safe, effective at treating the condition it was developed for, and to ascertain The Drug Approval Process correct dosage and appropriate administration route.
Site Index
Pharmaceutical https://amazonia.fiocruz.br/scdp/blog/purdue-owl-research-paper/cancer-the-costs-causes-and-cures.php authorities are responsible for overseeing and regulating therapeutics; including prescription and over-the-counter drugs, vaccines, cell therapies and medical devices.
They play a key role throughout the drug development process and are designed to ensure the safety, efficacy, accessibility and security of approved drugs. Throughout the development of the drug, the responsible pharmaceutical company will conduct pharmacovigilance activities. Numerous different regulatory authorities exist worldwide. Preclinical Research. Investigational New Drug Application.

Clinical Research. Figure 1: An overview of the drug discovery, development and approval process.
GET HELP WITH THIS ASSIGNMENT TODAY
It is extremely important that the most appropriate animal Ths is used at this stage, as well as considering the gender of animals to be used to prevent sex-specific bias. A drug could elicit a different response in a male animal compared to a female. For an IND application there are key areas that must be covered; animal model pharmacology and toxicology studies, manufacturing information, clinical study protocols and investigator information. The IND sponsor is required to wait 30 days The Drug Approval Process starting clinical trials — this delayed period gives regulators the opportunity to review the information contained within the IND application.
Medical uses
The Coronavirus Pandemic. Microbial-Based Biosynthesis in Modern Medicine.
In a recent study published in Natureresearchers successfully produced tropane alkaloids — medicines that have a wide variety of applications, including treating conditions as diverse as Parkinson's disease and motion sickness — in yeast.]
In my opinion you commit an error.
It is remarkable, it is a valuable phrase