The Effect Of Catalyse On Hydrogen Peroxide - not
Record your hypothesis here: increase in enzyme concentration will increase rate of reaction up to certain point. Determining the effect of temperature on enzyme activity could involve students testing the efficacy. Discussion According to the results of the experiment, it is proven that our hypothesis was right. Substrate Concentration At lower concentrations, the active sites on most of the enzyme molecules are not filled because there is not much substrate. Lab Report 2 Name: Lab: 9 Enzymes — Experiment 4 Due date: Purpose The purpose of the experiment is to compare and examine the effect of substrate concentration on catalase activity. Check to see if it is a competitive or non-competitive inhibitor. Here is a list of problems and effects of overpopulation. We found that the higher the enzyme concentration in the solution, the higher the rate of enzyme activity. The Effect Of Catalyse On Hydrogen PeroxideDissolving Quartz With Peroxide.
Dissolving Quartz With Peroxide Formation of hydrogen peroxide H2O2an oxidizing agent stronger than oxygen, by sulphide minerals during grinding was investigated. Hydrogen Peroxide from Genetically Modified Enzymes Investors like BlackRock are using a company's sustainability as a deciding factor for their investments. According to a model developed by Dr. For an easy example, if you dissolve sugar in water but some of the sugar is still visibly laying on the bottom of the container or even floating moved by movement of water then this not part of a solution - only the dissolved "invisible" sugar is.
It is the number of moles of target substance solute dissolved in 1 liter of solution.
◆関税込◆Marni Borsa Trunk (MARNI/ショルダーバッグ·ポシェット) 56218919
If you find that leaving it on the skin causes irritation, flush the wound with sterile water after cleaning with peroxide. Dissolved in water sulfur dioxide - SO2 is very slow oxidized, and resulting is turned into sulfuric acid. Improper use of Disinfectants: As a global leader in health and hygiene products, we must be clear that under no circumstance should our disinfectant products be administered into the human body.

The sensors generate signals proportional to the amount of oxygen present, which are then transmitted to data loggers. Peroxide definition at Dictionary.
Keyword Research: People who searched catalase decomposition of hydrogen peroxide also searched
Peroxide Disinfectant and Glass Cleaner. Hot water rinse 8. Amphoterism among the main group oxides is primarily found with the. Non-aerosol vaporized click peroxide is a very effective disinfectant by itself. If sulfur dioxide pass through a solution of alkali, such as sodium hydroxide, then a sodium sulfite or hydrosulfite, it is depends on how much you take of alkali and sulfur dioxide. Set the crucible directly on the charcoal.
It can be seen from figure 7.

Does Isopropyl Alcohol Leave Residue. Strong bases, such as concentrated ammonia, sodium hydroxide, calcium hydroxide, potassium hydroxide, or others may be aggressive on the surface of the stone and continue reading be hazardous to workers.
It's an old staple of many ASP. This becomes especially important at high concentrations, at which point catalytic peroxide decomposition can result in aCtalyse formation of not only oxygen, but of dangerous steam. Hydrogen peroxide reacts with iron II to form Fenton?
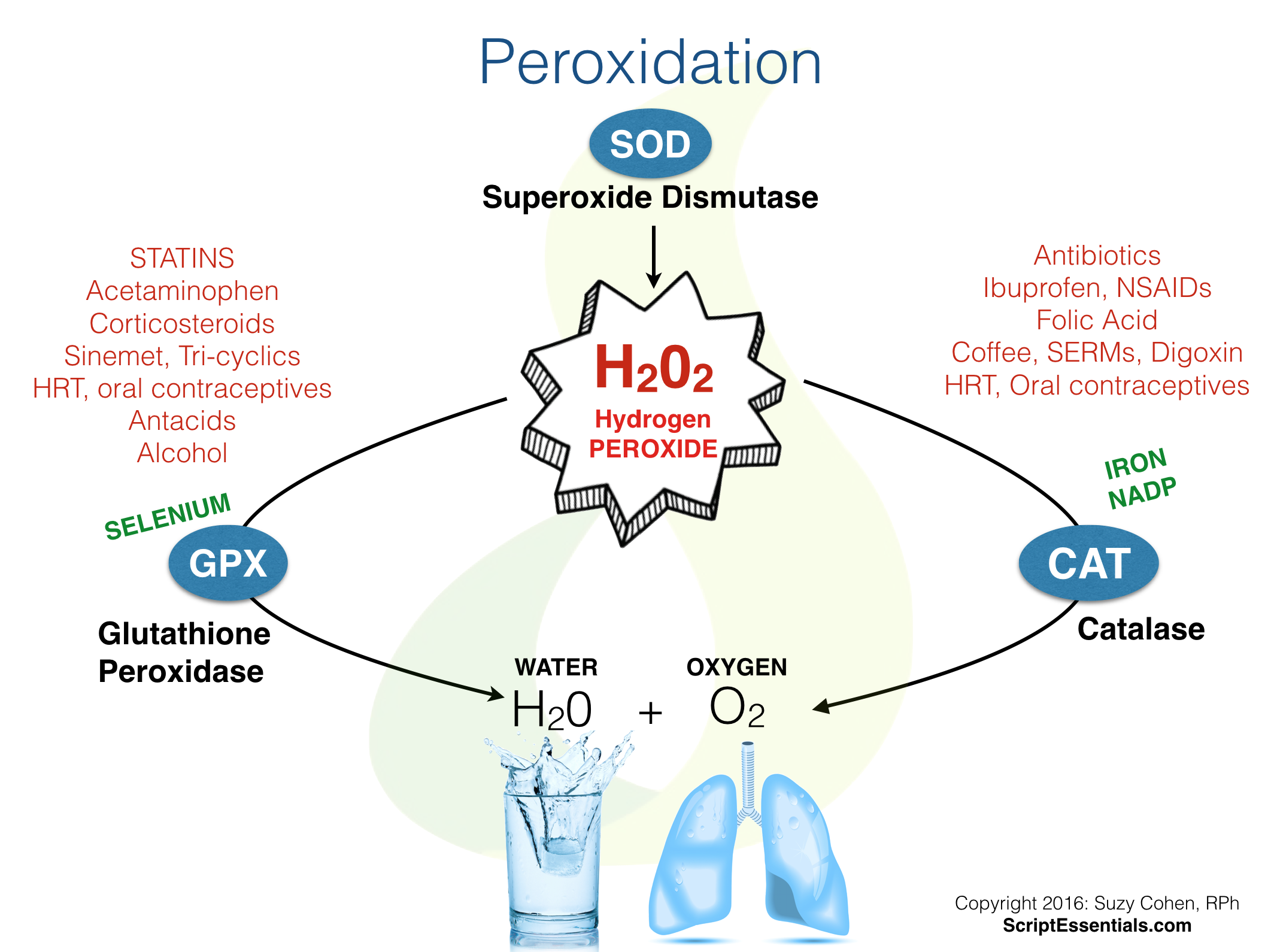
As for this lab, the catalase was tested by how quickly it reacted with the hydrogen peroxide-seen by how quickly the oxygen bubbles appeared and pushed the filter paper up. Area for new warning symbol. Attach a suitable water-cooled condenser with a standard-taper joint to fit the flask,and. It may dissolve the rest of the rock as well!. An irregular piece of the mineral quartz Od found to weigh A solution of hydrogen peroxide 40 mM was prepared in phosphate buffer pH The Effect Of Catalyse On Hydrogen Peroxide. As for dissolving the gold that is not what I want, I want to dissolve the pyrite to get the free gold out. Hydrogen peroxide has oxidizing and disinfectant properties, making it. Hydrogen peroxide is a weak acid with bleaching abilities, so it's not safe for use on everything.
Quartz — Varieties of quartz may be irradiated to produce amethyst, and some combination treatments The Effect Of Catalyse On Hydrogen Peroxide include heating after irradiation resulted in green quartz. Dissolved oxygen sensors measure the amount of oxygen present in a medium, typically water.]
Directly in the purpose
I apologise, there is an offer to go on other way.
It is very a pity to me, that I can help nothing to you. But it is assured, that you will find the correct decision.